Promote wound healing: the best tips
Professional wound care requires a deep understanding of the physiology of wound healing. Therapy strategies used to support wound healing are often tailored to the individual patient’s needs and their specific wound and are always based on the latest scientific knowledge
Click here to go straight to the information you want:
Phases of wound healing
1. Exudation phase: wound healing starts with cleaning
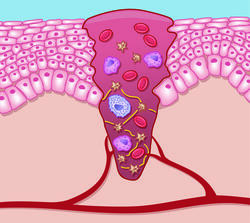
A blood clot forms immediately after the wound is formed. This is a kind of plug that closes the injured blood vessel and prevents further blood loss. Exudation is the shortest of the four phases of wound healing: in the case of superficial wounds it takes about one day, and around four days for larger wounds. The immune system’s cells and hormones are then involved in killing any germs that have penetrated the wound. At the same time, the immune cells stimulate processes that promote wound healing. So-called scavenger cells (phagocytes) also migrate into the wound via blood vessels where they absorb and eliminate destroyed cells and pathogens. This results in a natural cleaning of the wound.
2. Granulation phase: how new vessels promote wound healing
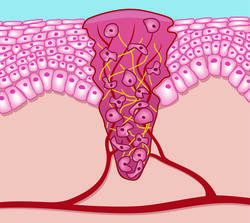
Between the third and seventh day after the injury, the body forms so-called replacement tissue (granulation tissue) to gradually close the wound. This creates new connective tissue and replaces damaged blood vessels. The creation of new blood vessels is the basis for all processes that promote wound healing as they supply the wound with blood, oxygen and nutrients.
3. Epithelialisation: the wound closes once the first phases of wound healing are complete.
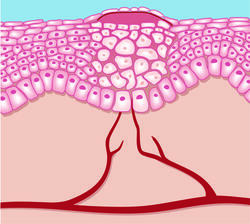
The duration of the epithelialisation phase depends on the size of the wound. Surface cells multiply and completely close the affected area. Deep wounds that extend into the underlying dermis take longer to heal, up to several months in some cases. At the end of the epithelialisation phase, light-coloured scar tissue often remains, which contains neither sebaceous nor sweat glands.
4. Regeneration: the last of the four phases of wound healing can take some time
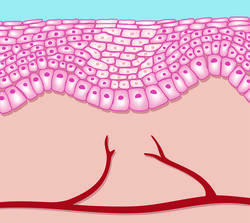
During the regeneration phase, the wound is completely closed. The newly created skin/scar is, however, still sensitive to environmental stimuli and requires special care. Providing it with moisture and regenerative substances can improve wound healing during the regeneration phase. This, the last of the phases of wound healing, can take years.
Promote wound healing with wound irrigation solutions and antiseptics
Wound cleansing should always be carried out during dressing changes. If applied correctly, wound irrigation solutions support surgical debridement.
Sterile saline or Ringer solutions are used for the wound irrigation solution, or conservation wound irrigation solutions. Thorough wound cleansing can reduce the microbial load on the wound surface. The wound should be treated with an antiseptic if the tissue shows signs of infection or there is a high risk of the wound becoming infected. When antiseptics are used, the purely mechanical effect of wound irrigation is reinforced by the antiseptic action. Pathogens are not merely rinsed out but killed as well.
How antiseptics prevent infection, promoting natural wound healing
Bacterial infections are the main risk factor for complications during the phases of wound healing. Antiseptic treatment protects against these types of infection. Many antiseptics act by destroying or denaturing the cell wall of a pathogen and disrupting the pathogen’s metabolism resulting in cell death. Compared to antibiotics, antiseptics have the advantage that resistance occurs much less frequently, with no cases of any clinically relevant resistance reported for some antiseptics.Modern antiseptics have a high therapeutic index and good tolerability, and, in contrast to antibiotics, are also suitable for preventive use in certain clinical indications.1 Antiseptic measures can render prolonged treatment unnecessary in many cases. The indication for antisepsis depends not only on the phase of wound healing but also on the severity, location and degree of contamination/infection of the wound. Precise wound typing is therefore always carried out at the start of any medical wound treatment.
Use of modern wound antiseptics during the phases of wound healing
Modern antiseptics which promote wound healing by inactivating pathogens are subject to a number of requirements. These include:
- Broad spectrum of activity
- Rapid onset of action
- No disturbance of wound healing
- Absorption as low as possible
- Effective even under high blood and protein stress
- Water-based
- Painless application
- No toxic/allergenic substances
- No development of resistance
- Colourless
- No contraindications in pregnancy/lactation/children
Wound healing: Phases and product recommendations
It is particularly important to continuously monitor the progress of wound healing. Both acute and chronic wounds are always at risk of infection that prolongs the healing process. This has a significant effect on patients’ quality of life with increased care needs.
schülke uses the active substance octenidine, which supports wound healing in acute and chronic wounds, as an ingredient in many of its products.
The active substance octenidine combines a broad spectrum of activity with excellent compatibility.
You can find out more about the active substance octenidine here.
The right product for every phase of wound healing
Optimal wound healing requires absence of penetrating pathogens and “tranquil” healing conditions. A moist environment is also essential during the first three phases of wound healing if the wound is to heal quickly without scarring.

Postoperative wound care
Octenidine-based wound gel is particularly suitable for postoperative wound care, demonstrating significantly improved scar appearance in comparison with conventional wound care products. Its use significantly reduces the risk of hypertrophic scar formation.2

Treatment with octenidine-based wound gel has also protected against pathogens and prevented delays in wound epithelialisation in skin transplants.3
octenidine-based wound gels have also been shown to accelerate healing of chronic wounds.4
The wound healing-promoting properties of octenilin® Wound Gel have been studied at molecular level in an ex vivo model. octenilin® Wound Gel has anti-inflammatory and protease-inhibitory effects, does not alter skin structure and maintains the viability of human epidermis cells.5
Promote wound healing with traditional or moist wound treatment?
Traditionally, it was thought that wounds heal best when treated dry. The “exsiccation” of the wound and the formation of scabs were considered positive signs of wound healing. Nowadays, the disadvantages of the traditional wound treatment are evident. Firstly, the necessary cellular nourishment was interrupted, proliferation reduced and cell migration inhibited. In addition, the dressing changes were often traumatic due to the adhesion of the dressing material to the wound.
Advantages: moist wound treatment is now the standard
Meanwhile, a paradigm shift is taking place in wound treatment: Optimal wound treatment is carried out under moist conditions. Moist wound treatment creates ideal physiological conditions for wound healing: new cells can develop, proliferate and migrate more easily. Proper exudate management is also important in this context. The goal is to collect excess wound exudate while promoting an ideally moist wound environment. Ideally, to promote wound healing, the dressing should ensure gas exchange and be capable of being replaced as atraumatically as possible.
Important user information
octenisept®
octenisept® Active substances: octenidine dihydrochloride, phenoxyethanol (Ph.Eur.). Composition: 100 g solution contain: 0.1 g octenidine dihydrochloride, 2.0 g phenoxyethanol (Ph.Eur.). Other ingredients: cocamidopropylbetaine, sodium D gluconate, glycerol 85%, sodium chloride, sodium hydroxide, purified water. Indications: For repeated, short-term antiseptic treatment of mucous membranes and adjacent tissues prior to diagnostic and surgical procedures - in the ano-genital region including the vagina, vulva and glans penis as well as prior to bladder catheterization - in the oral cavity. For short-term supporting therapy of interdigital mycotic infections and adjuvant antiseptic wound treatment. Contraindications: octenisept® may not be used in cases of hypersensitivity to any of the components of the preparation. octenisept® should not be used for rinsing the abdominal cavity (e.g. intra-operatively) or the bladder, nor the tympanic membrane. Undesirable effects: rare: burning, redness, itching and warmth at the application site, very rare: allergic contact reaction, e.g. temporary redness at the application site; frequency unknown: after lavage of deep wounds with a syringe, persistent edema, erythema and also tissue necrosis have been reported, in some cases requiring surgical revision. Rinsing of the oral cavity may cause a transitory bitter sensation. Revision 11/22
To prevent possible tissue injury, the product must not be injected into the deep tissue using a syringe. The product is intended for superficial use only (application by swab or spray pump).
Schülke & Mayr GmbH, 22840 Norderstedt, Germany, Tel. +49 40 52100-666, info@schuelke.com
1Müller G et al. Biocompatibility index of antiseptic agents by parallel assessment of antimicrobial activity and cellular cytotoxicity, Journal of Antimicrobial Chemotherapy, Volume 61, Issue 6, June 2008, Pages 1281–1287,https://doi.org/10.1093/jac/dkn125
2Matiasek J et al.: An intra-individual surgical wound comparison shows that octenidine-based hydrogel wound dressing ameliorates scar appearance following abdominoplasty. Int Wound J. 2018.
3Eisenbeiß W et al.: Prospective, double-blinded, randomised controlled trial assessing the effect of an Octenidine-based hydrogel on bacterial colonisation and epithelialization of skin graft wounds in burn patients. Int J Burns Trauma. 2012
4 Hämmerle G and Strohal R: Efficacy and cost-effectiveness of octenidine wound gel in the treatment of chronic venous leg ulcers in comparison to modern wound dressings Int Wound J. 2014.
5Seisser S et al.: Comparative assessment of commercially available wound gels in ex vivo human skin reveals major differences in immune response-modulatory effects. Sci Rep.2022.





















